|
Back to Blog
Ground state electron configuration6/23/2023  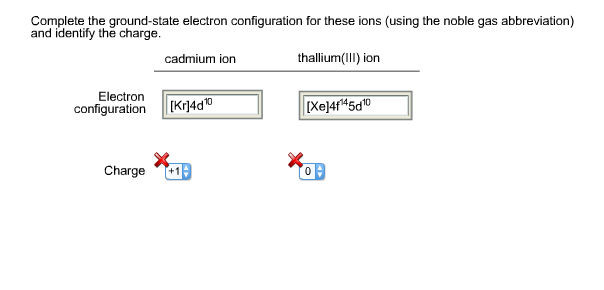
It can hold 14 electrons (2 electrons each orbit). The s orbital is the first orbital around the nucleus.Number of orbitals x 2 electrons = number of total electrons in the orbital.įor each principal quantum number, n, there is: This can be calculated with a simple formula: The number of electrons can be increased depending on the orbital size. The orbitals have different shapes and orientations. The circles around the nucleus where the electrons revolve in the atom are called the orbitals. 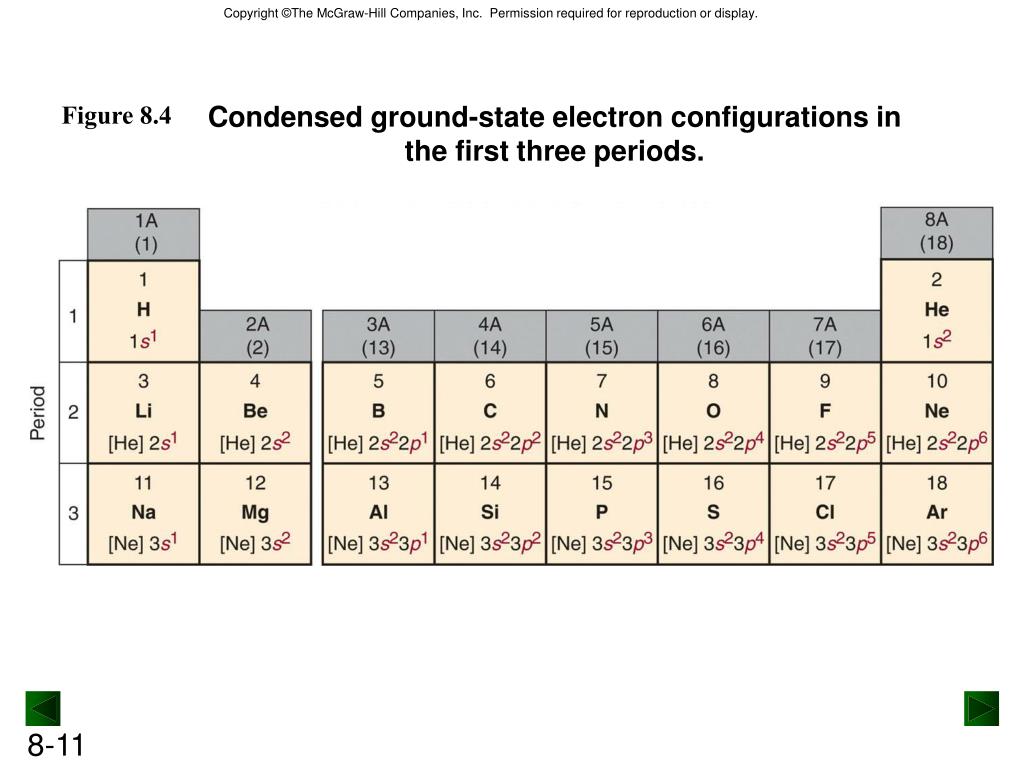
Core Electrons: Core electrons are chemically static in normal reaction conditions and are filled with inner energy levels.Valence Electrons: they travel farthest from the nucleus.Excited Electron state: The electron configuration of an atom at the highest energy possible.Ground Electron state: The electron configuration of an atom at the lowest energy possible.An essential part of every atom which carries the electricity. What are Electrons?Įlectron is a sub-atomic particle with a negative charge. Understanding the Ground state electron configuration leads to understanding atomic structure and the periodic table which is the base of everything in chemistry. The ground-state electron configuration enlightens many different atomic properties from the electrons present in an atom to the different orbital and hybridization. It is the base to understand the molecular bonding, structures, and properties. We shall focus on The Ground state electron configuration. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
